|
Back to Annual Meeting
Hemangioma Stem Cells Require Notch3 for Normal Hemangioma Formation in a Mouse Model
Naikhoba Munabi, B.A., Ryan W. England, B.S., Krista Hardy, B.S., Jan K. Kitajewski, PhD, Carrie J. Shawber, PhD, June K. Wu, M.D..
Columbia University, New York, NY, USA.
BACKGROUND:
Infantile hemangiomas (IHs) are the most common benign tumor of infancy, yet their pathogenesis remains poorly understood. IHs follow a known progression from an initial proliferative phase where hemangioma stem cells (HemSCs) predominate, through to an involution period with increased prevalence of hemangioma endothelial cells (HemECs). Our work has previously shown both in vitro and in vivo that Notch3, a mural cell-associated Notch gene, is expressed at high levels in HemSCs compared to HemECs. We thus hypothesize that downregulation of Notch3 expression is a critical and sufficient step for inducing progression in the natural history of IH.
METHODS:
Notch3 gene expression was inhibited in HemSCs using lentiviral expression of Notch3 shRNA (N3KO) vector with a scrambled lentivirus (scr) serving as control. Knockdown was confirmed with real-time quantitative PCR (RT-qPCR). Notch3 knockdown cells (HemSC-shN3) and control cells (HemSC-scr) were suspended in matrigel and injected subcutaneously in immunodeficient mice. Ultrasound (US) doppler imaging of matrigel plugs was performed weekly to assess for the presence of flow or vascular channels. After 3 weeks, the matrigel plugs were harvested and paraffin embedded, followed by histological staining of select sections. At 20X high-powered field magnification, samples were examined for vascular density, vessel diameter, number of adipocytes, and overall cellularity.
RESULTS:
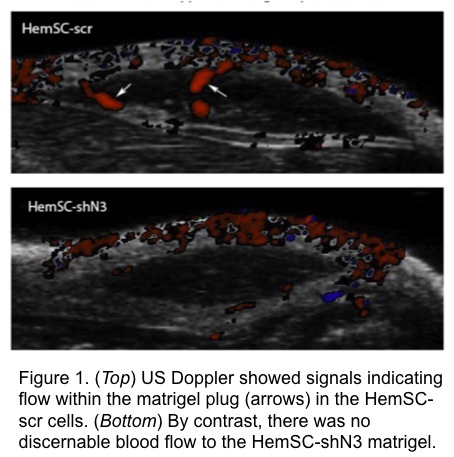
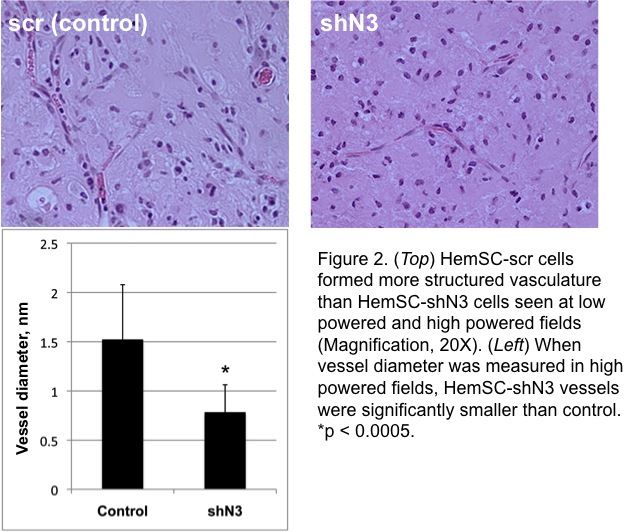
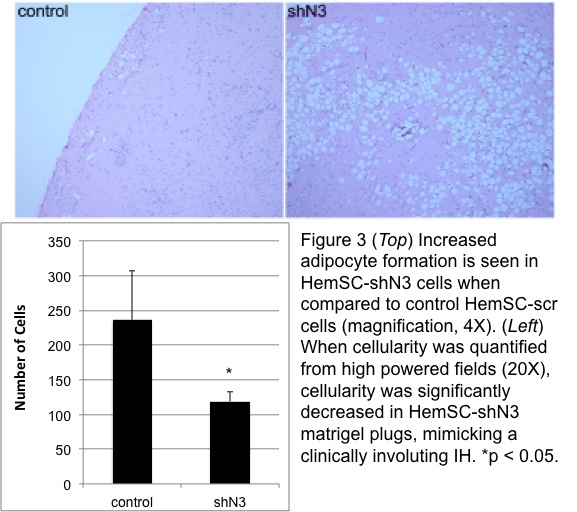
RT-qPCR confirmed transduction of Notch3 shRNA in HemSC with 50% decreased expression compared to control. US doppler of matrigel plugs detected the presence of blood flow within control HemSC-scr cells at as early as 2 weeks after implantation; in contrast, there was a sustained absence of discernable flow to the HemSC-shN3 matrigel plug (Fig 1). On histological examination, control IHs showed larger caliber blood vessels than HemSC-shN3, which was statistically significant (p < 0.0005, Fig 2). Overall cellularity of the HemSC-shN3 matrigel was statistically reduced when compared to control cells (p < 0.05), despite the presence of an increase in adipocyte formation (Fig 3).
CONCLUSIONS:
Knockdown of Notch3 expression in HemSCs leads to loss of doppler-detectable blood flow, decreased vessel caliber, cellularity, and increased adipocyte formation in a recapitulated mouse model of IH. The constellation of features seen in Notch3 knockdown cells imitates the clinical features of involuting IHs. These data suggest that Notch3 suppression may accelerate involution and can be a potential therapeutic target for IH.
Back to Annual Meeting
|









