Back to 2015 Annual Meeting
Thy1 Dependent Myofibroblast Differentiation in Radiation-Induced Capsular Contracture
Trevor C. Hansen, BS, Peter F. Koltz, MD, Collynn Woeller, PhD, Richard P. Phipps, PhD, Howard Langstein, MD.
University of Rochester, Rochester, NY, USA.
BACKGROUND: Pathologic scarring confers significant post-operative morbidity in implant-based breast reconstruction patients, notably in the form of capsular contracture. With the addition of radiation therapy, the incidence of this poorly understood complication approaches 100%. We have previously shown that Thy1 (formally called CD90), a glycophosphatadyl inositol membrane anchored glycoprotein, is important in myofibroblast differentiation and thus scar tissue formation. Herein, we characterize the expression of Thy1 in capsular scar tissue and test its ability to function as a potential pharmacologic target to modulate scar formation.
METHODS: Peri-implant scar samples from skin and both irradiated and non-irradiated breast capsules were analyzed using semi-quantitative RT-PCR for relative Thy1 mRNA expression. Capsular fibroblasts explanted from these primary tissues were grown in vitro and subjected to: treatment with pro-fibrotic cytokine transforming growth factor-β (TGFβ), transduction with a lentiviral vector designed to down-regulate Thy1 expression, and treatment with a novel Thy1 small molecule inhibitor: salinomycin (SNC). Resultant cells were analyzed for expression of Thy1, α-smooth muscle actin (αSMA), and collagen I.
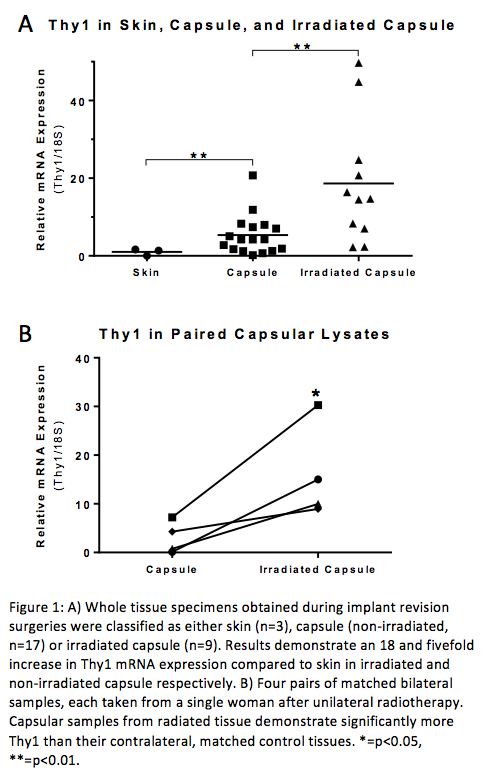
RESULTS: Relative to skin, a non-pathologic fibroblast control, non-irradiated and irradiated capsules expressed over five and 18 fold higher Thy1 mRNA levels respectively (p=0.0046 and 0.0041). Irradiated capsule Thy1 was also significantly increased over non-irradiated capsule (p=0.02). Furthermore, we obtained bilateral samples from four individuals who underwent unilateral radiation treatment, and discovered appreciably higher Thy1 levels in the radiated breast compared to the contralateral, non-irradiated breast (p=0.0468). In vitro, lentiviral-targeted knockdown of Thy1 not only decreased Thy1 by approximately 80%, but also significantly decreased αSMA and collagen protein expression. Furthermore, whereas treatment of explanted fibroblasts with TGF-β alone induced significant increases in αSMA and collagen levels, co-treatment with SNC resulted in protein expression at or below baseline.
CONCLUSIONS: Increased Thy1 in fibroblasts from irradiated breast capsules suggests a possible mechanism for the increased incidence of capsular contracture in radiotherapy-treated patients. That this elevation is seen when using the patient's own contralateral, non-irradiated breast as a control provides further evidence for the role of radiation in increasing loco-regional Thy1 levels. In vitro induction of fibroblast scar production can be ameliorated by reducing Thy1 expression using SNC. Targeted Thy1 inhibition using lentiviral siRNA also reduces scar component production, suggesting that SNC's effect may be mediated predominantly through Thy1 suppression. Given further understanding of Thy1's role in myofibroblast differentiation and scar production, molecular targeting could potentially reduce capsular contracture following radiation. Future studies on the effects of Thy1 modulation, including in vivo work, will be reported in the near future.
Back to 2015 Annual Meeting
|




