Optimizing Radiation Delivery in a Murine Model of Radiation-Induced Peri-Prosthetic Capsular Fibrosis
Alexandra J. Lin, BA1, Sarah Karinja, BA1, Julia Jin, BS1, Jaime Bernstein, BS1, Yoshi Toyoda, BA1, Omer Kaymakcalan, MD1, Jason A. Spector, MD FACS1, Jason A. Spector, MD FACS2.
1Laboratory for Bioregenerative Medicine & Surgery, Weill Cornell Medical College, New York, NY, USA, 2Nancy E. and Peter C. Meinig School of Biomedical Engineering, Cornell University, Ithaca, NY, USA.
BACKGROUND: Capsular contracture (CC) following breast reconstruction is a morbid complication, occurring in 5-10% of reconstructed patients, and in an even higher 40% of patients undergoing post-operative radiotherapy. Although it is well known that radiation increases rate of CC, there are few well-established animal models that faithfully replicate standard of care clinical practice, i.e. tissue expander placement at the time of mastectomy followed by delayed radiotherapy, weeks to months post-operatively. In an effort to better imitate current clinical practice, we locally irradiated implant sites 7 days post-operatively, rather than at the time of surgery. We also utilized the X-RAD 225Cx microirradiator (Precision X-ray) to deliver precisely dosed, implant-focused irradiation, more closely mimicking human radiotherapy. Lastly, as the optimal irradiation dose to induce murine CC is unknown, we assessed the effects of different RT doses on capsule thickness.
METHODS: Fifteen C57Bl/6 mice were divided into four treatment groups, differing in irradiation dose: 0 Gray (Gy), 10 Gy, 20 Gy, 30 Gy or 40 Gy. Hemispherical implants were created from polydimethylsiloxane and implanted subcutaneously into the mice dorsa. In one mouse per group, dosimeters were placed subcutaneously to allow for accurate measurement of the irradiation dose delivered. Seven days post-operatively (equivalent to approximately 8-12 weeks in humans), mice were locally irradiated with a single dose per implant of focused irradiation utilizing the X-RAD 225Cx microirradiator (Precision X-ray) with 3-dimensional treatment planning system software (Metropolis) operated by an experienced technician. After 21 days (30 days post-operatively), the mice were sacrificed.
RESULTS: Accurate radiation dosing was confirmed by the dosimeter readings. Localized radiation delivery was evaluated on physical exam and histology. Physical examination of skin overlying the implants demonstrated evidence of local radiation delivery, including circular patterns of hair blanching and thinning directly over the implants. Histologic analysis of the irradiated epidermis demonstrated radiation changes including keratin whorls and patches of uneven epidermal thickness, changes not present in adjacent non-irradiated epidermal regions. Furthermore, the effects of different doses on capsule thickness were assessed. Mice treated with 30 and 40 Gy doses experienced shortness of breath and coagulopathy, dying within 2 weeks of radiotherapy and suggesting that despite local delivery, 30-40 Gy can induce radiation poisoning in this murine model. There was no statistically significant difference in capsule thickness between the 0, 10 and 20 Gy groups.
CONCLUSIONS: Despite using sophisticated treatment-planning software and precise irradiation delivery, we were unable to demonstrate increased peri-prosthetic capsule thickness with localized irradiation, irrespective of dose. These results differ from those previously published, which demonstrated increased capsule thickness with irradiation. Given the evidence of local radiation delivery (dosimeter reading, physical exam findings, skin radiation changes), we believe the lack of increase in capsule thickness observed in our experiment is a real phenomenon, which may be model-specific. These "negative" data demonstrate the difficulty in translating the findings derived from animal models into clinical practice. 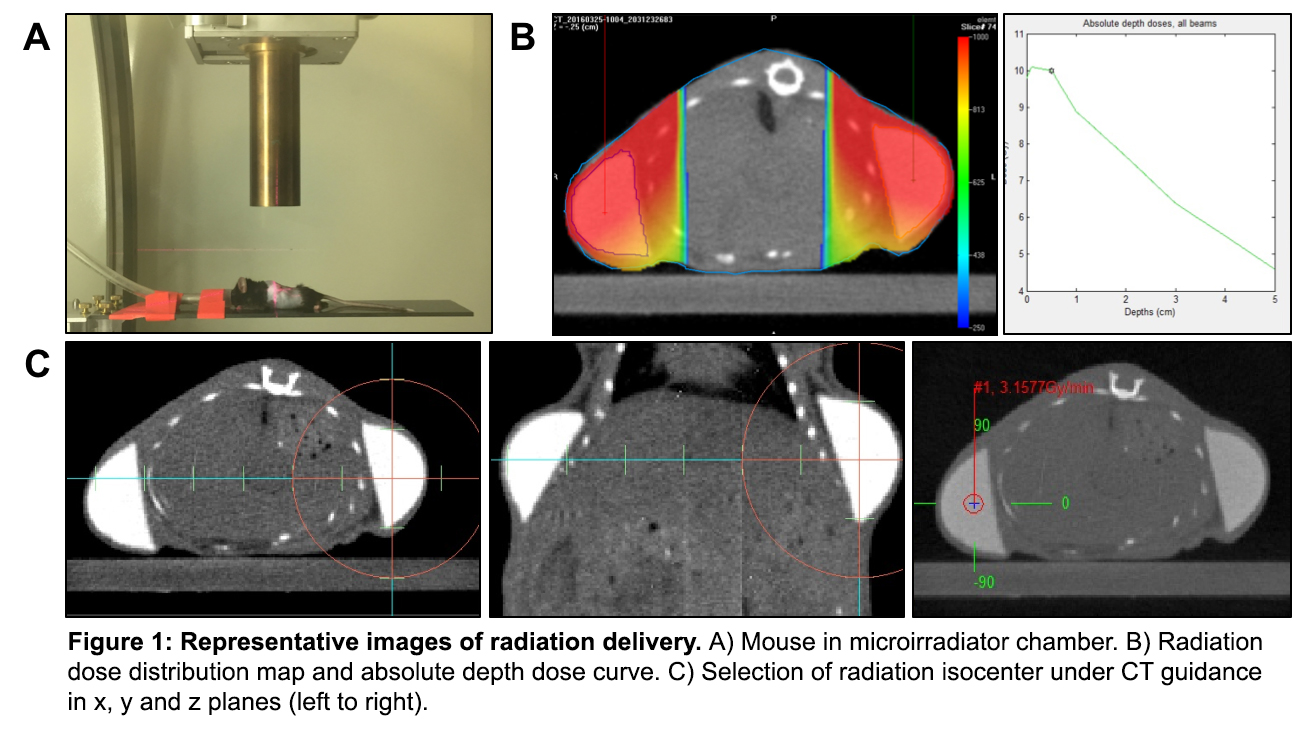
Back to 2017 Program




