Smoking May Have a Different Risk Factor Profile for Postoperative Complications in Plastic and General Surgical Procedures; A Propensity-Matched Analysis of 294,903 Patients from the NSQIP Database from 2005-2014
Rose Fu, MD1, Yoshiko Toyoda, BA1, Lu Li, MS1, Christine Rohde, MD, MPH, FACS2, David Otterburn, MD, FACS1.
1Weill Cornell Medical Center, New York, NY, USA, 2Columbia University Medical Center, New York, NY, USA.
Background
Smoking is an established risk factor for postoperative complications including poor wound healing. This modifiable risk factor is especially relevant in elective plastic surgical (PS) cases compared with urgent general surgical (GS) procedures. From 2005-2014, the U.S. adult smoking rate decreased from 20.9% to 16.8%. This study aims to investigate (1) whether smoking prevalence in the PS and GS patients paralleled national trends and (2) whether the postoperative complication profiles differ in PS and GS procedures when smoking is isolated as an independent risk factor.
Methods
The American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database was used to examine smoking prevalence and 30-day postoperative complications in patients who underwent PS and GS procedures. Common plastic surgical procedures where extensive planes of dissection make wound healing a greater concern and common general surgical procedures were selected for comparative analysis. Patients were propensity matched for demographics and comorbidities to isolate smoking as a risk factor.
Results
A total of 294,903 (36,454 PS and 258,449 GS) patients from 2005-2014 were examined. The smoking rates in GS cohort paralleled national trends (R=-0.85). Smoking rates in PS patients were lower than the national census for all years studied (p<0.01) (Figure). After propensity matching, GS smokers continued to be more comorbid compared to respective nonsmokers, with greater incidences of diabetes, hypertension, dypsnea, and prior cardiac surgery (p<0.01). After propensity matching, PS smokers were not significantly different than nonsmokers in demographics or comorbidities. Smokers had increased rates of superficial surgical site infections (SSI) (p<0.01), PE (p<0.01), and MI (p<0.02) for GS cohort, but not for PS cohort. Both PS and GS smokers had increased incisional dehiscence (p<0.01), deep SSI (PS: p=0.01, GS: p<0.01), and reoperation (p<0.01). Further analysis based on duration of smoking found that patients with ≥11 pack-years experienced significant increases in incidences of deep SSI (PS: p=0.02, GS: p=0.02) and reoperation (PS: p=0.05, GS: p<0.01). In GS smokers only, ≥ 21 pack-years was associated with increased sepsis (p<0.01), MI (p=0.04) and organ/space SSI (p<0.01), and ≥ 31 pack-years was associated with increased incisional dehiscence (p<0.01). Bleeding, graft failure, and deep vein thrombosis rates were not significantly different between smokers and nonsmokers in either PS or GS cohort.
Conclusions
This is the first propensity-matched, large-scale database analysis isolating smoking as a risk factor for postoperative complications in PS and GS procedures. The contrast in smoking rates between PS and GS patients may highlight differences in the patient population seeking
these procedures as well as physicians' patient selection for elective procedures. Our data indicates that smoking is an independent and a dose-dependent risk factor in both PS and GS procedures. Finally, these results suggest smoking may have different risk factor profiles for postoperative complications in PS versus GS patient populations emphasizing the need for caution when generalizing general surgical evidence for use in the plastic surgical population. 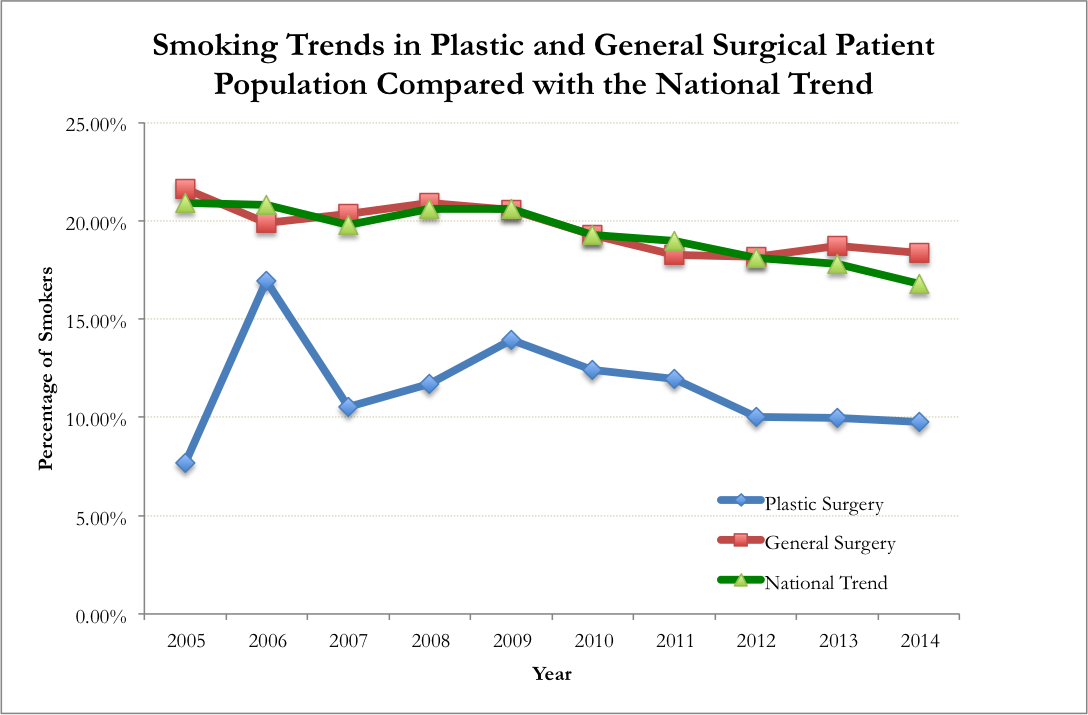
Back to 2017 Program




